Tai W, Yang K, Liu Y, Li R, Feng S, Chai B, Zhuang X, Qi S, Shi H, Liu Z, Lei J, Ma E, Wang W, Tian C, Le T, Wang J, Chen Y, Tian M, Xiang Y, Yu G, Cheng G.
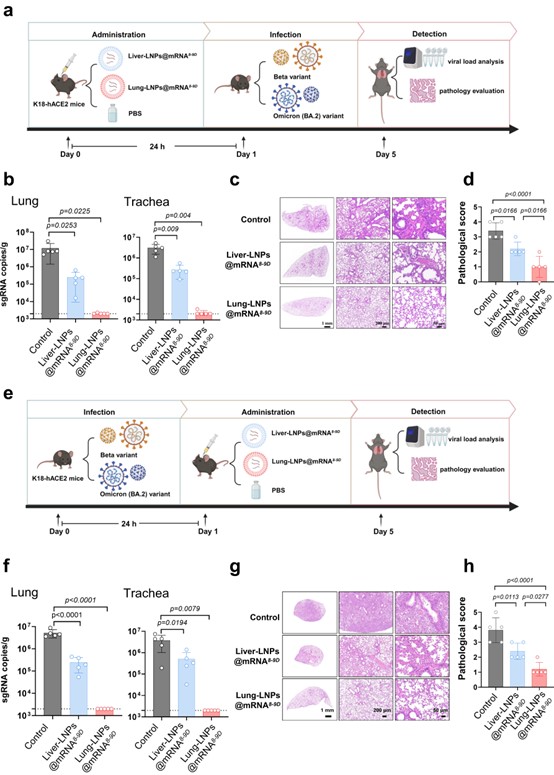
The respiratory system, especially the lung, is the key site of pathological injury induced by SARS-CoV-2 infection. Given the low feasibility of targeted delivery of antibodies into the lungs by intravenous administration and the short half-life period of antibodies in the lungs by intranasal or aerosolized immunization, mRNA encoding broadly neutralizing antibodies with lung-targeting capability can perfectly provide high-titer antibodies in lungs to prevent the SARS-CoV-2 infection. Here, we firstly identify a human monoclonal antibody, 8-9D, with broad neutralizing potency against SARS-CoV-2 variants. The neutralization mechanism of this antibody is explained by the structural characteristics of 8-9D Fabs in complex with the Omicron BA.5 spike. In addition, we evaluate the efficacy of 8-9D using a safe and robust mRNA delivery platform and compare the performance of 8-9D when its mRNA is and is not selectively delivered to the lungs. The lung-selective delivery of the 8-9D mRNA enables the expression of neutralizing antibodies in the lungs which blocks the invasion of the virus, thus effectively protecting female K18-hACE2 transgenic mice from challenge with the Beta or Omicron BA.1 variant. Our work underscores the potential application of lung-selective mRNA antibodies in the prevention and treatment of infections caused by circulating SARS-CoV-2 variants.